SCFAs Part 3: Decrypting the butyrate paradox: can excess butyrate be toxic?
Several supplement companies now offer various forms of oral butyrate supplements. But should we really be taking butyrate? Can too much butyrate be a bad thing?
In part 2 of the short-chain fatty acid (SCFA) series, I discussed the many benefits of the four-carbon microbial metabolite butyrate on host physiology. Thanks to its anti-inflammatory, immunoregulatory, and barrier-protective qualities, butyrate has received quite a lot of attention in the self-help, functional, and integrative medicine communities. And if you search PubMed, you’ll find dozens of studies showing the benefits of butyrate. Thus, it’s no surprise that supplement companies now offer oral butyrate supplements in various forms.
However, if you dig a bit deeper, you’ll also find several studies that suggest that butyrate is paradoxical; i.e. low concentrations of butyrate are beneficial, but high concentrations can be harmful, particularly in those with a compromised gut. Unfortunately, this hasn’t been talked about much on the blogosphere or the literature, and I think it’s important to have a nuanced discussion on butyrate. In this article, I’ll break down several studies that shed light on the butyrate paradox. First though, I’ll give a brief overview of gut anatomy, as it’s crucial to understanding butyrate’s effect on the gut.
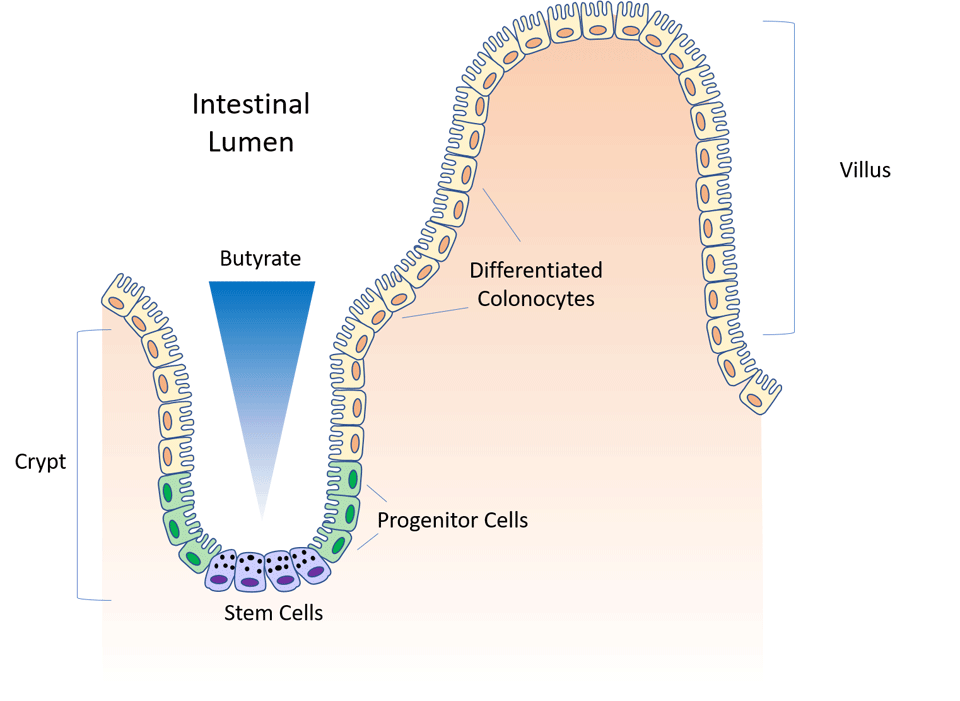
Gut anatomy 101: crypt and villus
The gut epithelium undergoes continuous renewal throughout life.1 Invaginations in the intestinal wall form crypts, while outward protrusions form villi (singular: villus). You can think of the crypt as a “valley” and the villus as the “mountain”. Stem cells lie at the bottom of the crypt and periodically produce progenitor cells, which in turn differentiate into mature colonocytes (colon cells) as they move up the villus. This replenishes older cells which slough off at the top. Through this process, the entire gut epithelium turns over roughly every 4-5 days.2
Intestinal crypts protect stem cells from excess butyrate
Despite knowledge of the existence of crypts for centuries, the reason for the structure of crypts was largely unknown until very recently. In 1974, Cheng and Leblond hypothesized that the crypt might offer rapidly dividing stem and progenitor cells protection from pathogens and toxins present in the intestinal lumen.3
In 2016, a Washington University research group found evidence to support this hypothesis. Using an innovative high throughput approach, they screened various microbial metabolites for their ability to suppress proliferation in cultured stem cells. To their surprise, they found that of all the metabolites, butyrate was the most potent inhibitor of stem and progenitor cell proliferation.4
In a living organism, however, butyrate did not cause this suppression. They wondered if butyrate was not being metabolized by stem cells at the bottom of the crypt. To find out, they used butyrate containing radiolabeled 13C to assess the rate of butyrate metabolism in various locations. They found a 3-fold higher level of 13C-labeled acetyl CoA (an endproduct of butyrate oxidation) in cells at the top of the crypt compared to stem and progenitor cells at the base of the crypt. The authors summarized the findings:
“Taken together, these data suggest that differentiated colonocytes located at the top of crypts can metabolize butyrate as an energy source, thus potentially preventing exposure of the stem cell niche to high levels of luminal butyrate.”
Butyrate prevents stem cell proliferation and wound repair in cases of mucosal injury
The researchers then turned to a mouse model of colitis, to understand how disruption of the crypt architecture might affect stem cell proliferation in the presence of butyrate. Suppression of stem cell proliferation would prevent the epithelial cell turnover required for healing mucosal injuries. They found that administering butyrate reduced epithelial proliferation in the crypts directly adjacent to colonic ulcers and delayed wound repair. In a separate experiment, they found that using antibiotics to knock down butyrate-producing microbes reduced ulcer size after a colitis insult. Reintroduction of butyrate or butyrate-producing organisms resulted in a return to large ulcers.
In a similar study, Zhang et al. introduced a human-derived butyrate-producing bacterium Anaerostipes hadrus into mice with and without colitis. In healthy mice, A. hadrus elevated levels of buyrate in the lumen, and had no adverse effects. In experimental colitis, mice receiving A. hadrus displayed increased disease activity and greater mortality.5
Another study found that supplementation with the soluble fiber inulin, known to stimulate the production of butyrate, resulted in increased pro-inflammatory gene expression in the colon of IBD-prone (IL-10 knockout) mice. This did not occur in control, wild-type mice.6 Together, these studies suggests that the benefits of butyrate, butyrate producers, and even potentially fermentable fibers, depends on the condition of the gut. These data may also partly explain why some inflammatory bowel disease (IBD) patients seem to improve upon reducing FODMAP intake.7
Low abundance of butyrate producers: cause or consequence?
For years, researchers have observed that patients with IBD have reduced levels of butyrate-producing bacteria8–10. It has been thought that this is a potential cause of disease. While this lack of butyrate producers is certainly representative of dysbiosis, this research outlined above makes me wonder if a lack of butyrate producers is actually a consequence of the disease. In other words, is it possible that an inflamed gut that is not in the condition to tolerate luminal butyrate might select against butyrate producers to protect stem cells and allow proliferation and mucosal repair?
After reviewing the data and having these questions come up in my head, I admit I was surprised by the author’s interpretations of their own findings in the discussion:
“Although [butyrate] negatively affects wound repair in the short term, it may actually benefit the host in the long-term. By suppressing the rapid expansion of stem cells following mucosal damage of the epithelium, butyrate may prevent stem cells from dividing while in direct contact with genotoxic luminal contents.”
While this is certainly possible, I think this may be representative of what often happens in science: we get caught up in our own hypotheses and what we think we know to be true. Butyrate has been considered beneficial for quite some time, so it’s hard to wrap our heads around a new paradigm: one in which butyrate could be detrimental in cases of impaired gut integrity. Of course, it’d be nice to have more than just these few studies to rely on. So let’s look at a few others…
Ulcerative colitis patients have impaired butyrate oxidation
Evidence from in vitro and in vivo studies suggests that oxidation of butyrate is impaired in ulcerative colitis (UC). De Preter et al. found that the average butyrate oxidation rate in UC biopsies was reduced to almost half that of the control biopsies. (When stratified based on severity, butyrate oxidation rate in inactive and mild disease was no different than controls, but butyrate oxidation rate in moderate and severe disease was significantly reduced. Interestingly, there was also a huge variability in butyrate oxidation among controls.)11
When you look at studies of butyrate kinetics, it becomes abundantly clear that the concentration of butyrate is not the problem. For both controls and UC biopsies, increasing luminal butyrate concentrations beyond 1mM did not increase butyrate oxidation. It seems that, beyond the 1 mM saturation point, the concentration of butyrate is not the limiting factor. The authors write:
“[…] local application of higher concentrations of butyrate does not overcome the mucosal energy deficiency in UC and […] stimulation of butyrate production through indigestible carbohydrates or butyrate enemas may not be effective in the treatment of this disease.” 11
In a follow-up study, Thibault et al. showed that the decreased butyrate oxidation is a result of reduced butyrate uptake by the inflamed mucosa. Proinflammatory cytokines downregulate expression of MCT1, a transporter of many molecules, including butyrate.12 Kovarik et al. also showed that butyrate has a reduced anti-inflammatory effect in IBD patients.13
Clinical trials, animal studies, and cell culture experiments show mixed results
Indeed, animal studies and human clinical trials have had contradictory results when butyrate enemas or butyrate treatment were tested in ulcerative colitis.
Butyrate enemas in humans:
A beneficial effect of butyrate enemas was found in two small non-placebo controlled studies14,15 in addition to two controlled crossover studies16,17 One small randomized placebo-controlled study in patients with mild to moderate colitis found a beneficial effect of SCFA enemas containing acetate, propionate, and butyrate. 18 However, the largest randomized placebo-controlled trial to date (91 patients) found no therapeutic value of SCFA enemas for colitis, though patients with milder, short episodes of colitis were slightly more likely to benefit19. Another large, randomized placebo-controlled trial (38 patients) found no significant difference between patients receiving saline placebo and butyrate enemas. In fact, 9 of 19 patients saw clinical improvement with the saline enema, while only 7 of 19 patients improved with the butyrate enema over the 6 week study.20 Granted, none of these studies showed an explicit harmful effect of butyrate, but I’d venture to guess that the researchers did not expect one and therefore were not looking for signs of potential mucosal toxicity.
As far as I know, no randomized controlled trials have been done on oral butyrate supplementation in humans.
Animal studies:
What about animal studies? One study found that oral sodium butyrate attenuated colitis in mice.21 However, high concentration butyrate enemas have also been found to induce apoptosis and UC-like lesions in mice22. In a mouse model of colorectal cancer, both inulin and intrarectal butyrate have both been shown to transform the colonic epithelium and increase the formation of tumors.23,24
In mice, chronic orally administered SCFAs induced T cell-mediated ureter tissue inflammation, leading to kidney hydronephrosis, a backup of fluid in the kidney. From the abstract:
“SCFAs, when systemically administered at levels higher than physiological levels, cause dysregulated T cell responses and tissue inflammation in the renal system.” 25
Cell culture:
Notably, several studies have now shown that SCFAs can promote either immune regulation or immune activation, depending on the inflammatory milieu.26,27 In other words, in a state of quiescence, SCFAs promote immune regulation; in a state of inflammation, they promote amplification of the immune response.
Furthermore, butyrate administration at low concentration (2 mM) to a cultured model of an epithelium improves gut barrier integrity by accelerating assembly of tight junction proteins28,29 However, at high concentrations (8 mM), butyrate increases barrier permeability.28
Butyrate also promotes the expression of virulence factors in pathogenic E.coli. These E.coli thus tend to colonize the colon where levels of butyrate are highest.30
Should we ever consider butyrate supplementation?
Possibly. Anecdotally, there are many people who have found benefits of butyrate supplementation, and some patients in the trials I mentioned did see clinical improvement with butyrate enemas. Butyrate certainly has many benefits, as I discussed in part 2 of this series. However, there are currently no double-blind, placebo controlled trials of oral butyrate supplementation in humans that have been performed, so my current thinking is that we need a more nuanced, case-by-case approach.
If you have severe mucosal damage, such as highly active ulcerative colitis, I would tend to advise against butyrate enemas or supplementation. Butyrate might be worth considering for milder cases, for those in remission or with refractory colitis, or for people without GI conditions. Still, it’s important to avoid supra-physiological levels.31
Until recently, the most widely available form of butyrate has been butyrate salts, such as sodium butyrate or calcium/magnesium butyrate. However, butyrate salts may partially dissolve in the mouth. High concentrations of oral butyrate have been associated with periodontitis and may inhibit the proliferation of gingival epithelial cells.32,33
I’ve recently become aware of a new oral supplement formula from Tesseract called ProButyrate that may provide a way to administer lower doses of therapeutic butyrate in a sustained release manner. The molecular trap formulation from Tesseract is specifically targeted for release in the colon and prevents dissolution of butyrate in the oral cavity. It also reduces the dose required to achieve therapeutic effect, with a recommended dose of 600-1200 mg/day, compared to 3-4 g/day for most butyrate salts. I have no affiliation with Tesseract, but look forward to seeing clinical trials on these supplements in the future. If you decide to supplement with butyrate, I believe this is currently the best option available.
Prebiotics: a more physiological butyrate response?
Because of the nature of gut fermentation, prebiotic fibers are also likely to provide a slower and more dispersed release of butyrate in the colon. Our gut anatomy and gut ecology evolved on a diet rich in fermentable fibers, and this is the butyrate precursor that our guts are most accustomed to. Prebiotics also have many benefits beyond butyrate production, including anti-inflammatory effects on the gut epithelium, and the proliferation of beneficial microbes. Still, it’s important to note that even large quantities of prebiotics may be detrimental to healing in cases of severe gut inflammation and mucosal injury.
What If I get a stool test and I have low butyrate? Should I supplement?
Unfortunately, many stool tests that are clinically available use improper sample processing methodology for measuring SCFAs, and therefore cannot be trusted for their accuracy. Furthermore, fecal SCFA levels are not necessarily representative of luminal SCFA concentrations. Stay tuned for part 6 of the SCFA series, where I’ll be addressing this issue in detail.
Takeaways:
- Butyrate can suppress epithelial stem cell proliferation and repair. The normal crypt architecture protects stem cells from high amounts of butyrate exposure.
- Butyrate supplementation should be used with extreme caution in cases of severe gut inflammation and epithelial damage, particularly in active ulcerative colitis. High-dose probiotic and prebiotic supplementation should also be used with caution, as butyrogenic fibers or butyrate-producing bacteria could also potentially exacerbate colitis.
- Small doses of butyrate supplementation may be helpful in those with mild colitis or those without GI issues, but prebiotics likely provide butyrate in more physiologically normal concentrations.
That’s all for now. Next up in the series: acetate and metabolic syndrome!
Sources:
- Stappenbeck, T. S., Wong, M. H., Saam, J. R., Mysorekar, I. U. & Gordon, J. I. Notes from some crypt watchers: regulation of renewal in the mouse intestinal epithelium. Curr. Opin. Cell Biol. 10, 702–709 (1998).
- van der Flier, L. G. & Clevers, H. Stem cells, self-renewal, and differentiation in the intestinal epithelium. Annu. Rev. Physiol. 71, 241–260 (2009).
- Cheng, H. & Leblond, C. P. Origin, differentiation and renewal of the four main epithelial cell types in the mouse small intestine V. Unitarian theory of the origin of the four epithelial cell types. Am. J. Anat. 141, 537–561 (1974).
- Kaiko, G. E. et al. The Colonic Crypt Protects Stem Cells from Microbiota-Derived Metabolites. Cell 165, 1708–1720 (2016).
- Zhang, Q. et al. Accelerated dysbiosis of gut microbiota during aggravation of DSS-induced colitis by a butyrate-producing bacterium. Sci. Rep. 6, (2016).
- Kuo, S.-M., Chan, W.-C. & Hu, Z. Wild-type and IL10-null mice have differential colonic epithelial gene expression responses to dietary supplementation with synbiotic Bifidobacterium animalis subspecies lactis and inulin. J. Nutr. 144, 245–251 (2014).
- Gearry, R. B. et al. Reduction of dietary poorly absorbed short-chain carbohydrates (FODMAPs) improves abdominal symptoms in patients with inflammatory bowel disease-a pilot study. J. Crohns Colitis 3, 8–14 (2009).
- Willing, B. P. et al. A pyrosequencing study in twins shows that gastrointestinal microbial profiles vary with inflammatory bowel disease phenotypes. Gastroenterology 139, 1844–1854.e1 (2010).
- Sokol, H. et al. Low counts of Faecalibacterium prausnitzii in colitis microbiota. Inflamm. Bowel Dis. 15, 1183–1189 (2009).
- Machiels, K. et al. A decrease of the butyrate-producing species Roseburia hominis and Faecalibacterium prausnitzii defines dysbiosis in patients with ulcerative colitis. Gut 63, 1275–1283 (2014).
- De Preter, V. et al. Kinetics of butyrate metabolism in the normal colon and in ulcerative colitis: the effects of substrate concentration and carnitine on the β-oxidation pathway. Aliment. Pharmacol. Ther. 34, 526–532 (2011).
- Thibault, R. et al. Down-Regulation of the Monocarboxylate Transporter 1 Is Involved in Butyrate Deficiency During Intestinal Inflammation. Gastroenterology 133, 1916–1927 (2007).
- Kovarik, J. J. et al. Impaired anti-inflammatory efficacy of n-butyrate in patients with IBD. Eur. J. Clin. Invest. 41, 291–298 (2011).
- Breuer, R. I. et al. Rectal irrigation with short-chain fatty acids for distal ulcerative colitis. Dig. Dis. Sci. 36, 185–187 (1991).
- Steinhart, A. H., Brzezinski, A. & Baker, J. P. Treatment of refractory ulcerative proctosigmoiditis with butyrate enemas. Am. J. Gastroenterol. 89, 179–183 (1994).
- Scheppach, W. et al. Effect of butyrate enemas on the colonic mucosa in distal ulcerative colitis. Gastroenterology 103, 51–56 (1992).
- Lührs, H. et al. Butyrate inhibits NF-kappaB activation in lamina propria macrophages of patients with ulcerative colitis. Scand. J. Gastroenterol. 37, 458–466 (2002).
- Vernia, P. et al. Short-chain fatty acid topical treatment in distal ulcerative colitis. Aliment. Pharmacol. Ther. 9, 309–313 (1995).
- Breuer, R. I. et al. Short chain fatty acid rectal irrigation for left-sided ulcerative colitis: a randomised, placebo controlled trial. Gut 40, 485–491 (1997).
- Steinhart, A. H., Hiruki, T., Brzezinski, A. & Baker, J. P. Treatment of left-sided ulcerative colitis with butyrate enemas: a controlled trial. Aliment. Pharmacol. Ther. 10, 729–736 (1996).
- Vieira, E. L. M. et al. Oral administration of sodium butyrate attenuates inflammation and mucosal lesion in experimental acute ulcerative colitis. J. Nutr. Biochem. 23, 430–436 (2012).
- Ohkusa, T. et al. Induction of experimental ulcerative colitis by Fusobacterium varium isolated from colonic mucosa of patients with ulcerative colitis. Gut 52, 79–83 (2003).
- Pajari, A.-M. et al. Promotion of intestinal tumor formation by inulin is associated with an accumulation of cytosolic beta-catenin in Min mice. Int. J. Cancer 106, 653–660 (2003).
- Belcheva, A. et al. Gut microbial metabolism drives transformation of MSH2-deficient colon epithelial cells. Cell 158, 288–299 (2014).
- Park, J., Goergen, C. J., HogenEsch, H. & Kim, C. H. Chronically Elevated Levels of Short-Chain Fatty Acids Induce T Cell-Mediated Ureteritis and Hydronephrosis. J. Immunol. Baltim. Md 1950 196, 2388–2400 (2016).
- Kim, M. H., Kang, S. G., Park, J. H., Yanagisawa, M. & Kim, C. H. Short-chain fatty acids activate GPR41 and GPR43 on intestinal epithelial cells to promote inflammatory responses in mice. Gastroenterology 145, 396-406.e1–10 (2013).
- Park, J. et al. Short-chain fatty acids induce both effector and regulatory T cells by suppression of histone deacetylases and regulation of the mTOR-S6K pathway. Mucosal Immunol. 8, 80–93 (2015).
- Peng, L., He, Z., Chen, W., Holzman, I. R. & Lin, J. Effects of butyrate on intestinal barrier function in a Caco-2 cell monolayer model of intestinal barrier. Pediatr. Res. 61, 37–41 (2007).
- Peng, L., Li, Z.-R., Green, R. S., Holzman, I. R. & Lin, J. Butyrate Enhances the Intestinal Barrier by Facilitating Tight Junction Assembly via Activation of AMP-Activated Protein Kinase in Caco-2 Cell Monolayers. J. Nutr. 139, 1619–1625 (2009).
- Nakanishi, N. et al. Regulation of virulence by butyrate sensing in enterohaemorrhagic Escherichia coli. Microbiol. Read. Engl. 155, 521–530 (2009).
- Roda, A. et al. A new oral formulation for the release of sodium butyrate in the ileo-cecal region and colon. World J. Gastroenterol. 13, 1079–1084 (2007).
- Tsuda, H., Ochiai, K., Suzuki, N. & Otsuka, K. Butyrate, a bacterial metabolite, induces apoptosis and autophagic cell death in gingival epithelial cells. J. Periodontal Res. 45, 626–634 (2010).
- Ochiai, K. & Kurita-Ochiai, T. Effects of butyric acid on the periodontal tissue. Jpn. Dent. Sci. Rev. 45, 75–82 (2009).
- Burger-van Paassen, N. et al. The regulation of intestinal mucin MUC2 expression by short-chain fatty acids: implications for epithelial protection. Biochem. J. 420, 211–219 (2009).
- Hatayama, H., Iwashita, J., Kuwajima, A. & Abe, T. The short chain fatty acid, butyrate, stimulates MUC2 mucin production in the human colon cancer cell line, LS174T. Biochem. Biophys. Res. Commun. 356, 599–603 (2007).
SCFAs Part 3: Decrypting the butyrate paradox: can excess butyrate be toxic?
Several supplement companies now offer various forms of oral butyrate supplements. But should we really be taking butyrate? Can too much butyrate be a bad thing?
In part 2 of the short-chain fatty acid (SCFA) series, I discussed the many benefits of the four carbon -microbial metabolite butyrate on host physiology. Thanks to its anti-inflammatory, immunoregulatory, and barrier-protective qualities, butyrate has received quite a lot of attention in the self-help, functional, and integrative medicine communities. And if you search PubMed, you’ll find dozens of studies showing the benefits of butyrate. Thus, it’s no surprise that supplement companies now offer oral butyrate supplements in various forms.
However, if you dig a bit deeper, you’ll also find several studies that suggest that butyrate is paradoxical; i.e. low concentrations of butyrate are beneficial, but high concentrations can be harmful, particularly in those with a compromised gut. Unfortunately, this hasn’t been talked about much on the blogosphere or the literature, and I think it’s important to have a nuanced discussion on butyrate. In this article, I’ll break down several studies that shed light on the butyrate paradox. First though, I’ll give a brief overview of gut anatomy, as it’s crucial to understanding butyrate’s effect on the gut.
Gut anatomy 101: crypt and villus
The gut epithelium undergoes continuous renewal throughout life.1 Invaginations in the intestinal wall form crypts, while outward protrusions form villi (singular: villus). You can think of the crypt as a “valley” and the villus as the “mountain”. Stem cells lie at the bottom of the crypt and periodically produce progenitor cells, which in turn differentiate into mature colonocytes (colon cells) as they move up the villus. This replenishes older cells which slough off at the top. Through this process, the entire gut epithelium turns over roughly every 4-5 days.2
Intestinal crypts protect stem cells from excess butyrate
Despite knowledge of the existence of crypts for centuries, the reason for the structure of crypts was largely unknown until very recently. In 1974, Cheng and Leblond hypothesized that the crypt might offer rapidly dividing stem and progenitor cells protection from pathogens and toxins present in the intestinal lumen.3
In 2016, a Washington University research group found evidence to support this hypothesis. Using an innovative high throughput approach, they screened various microbial metabolites for their ability to suppress proliferation in cultured stem cells. To their surprise, they found that of all the metabolites, butyrate was the most potent inhibitor of stem and progenitor cell proliferation.4
In a living organism, however, butyrate did not cause this suppression. They wondered if butyrate was not being metabolized by stem cells at the bottom of the crypt. To find out, they used butyrate containing radiolabeled 13C to assess the rate of butyrate metabolism in various locations. They found a 3-fold higher level of 13C-labeled acetyl CoA (an endproduct of butyrate oxidation) in cells at the top of the crypt compared to stem and progenitor cells at the base of the crypt. The authors summarized the findings:
“Taken together, these data suggest that differentiated colonocytes located at the top of crypts can metabolize butyrate as an energy source, thus potentially preventing exposure of the stem cell niche to high levels of luminal butyrate.”
Butyrate prevents stem cell proliferation and wound repair in cases of mucosal injury
The researchers then turned to a mouse model of colitis, to understand how disruption of the crypt architecture might affect stem cell proliferation in the presence of butyrate. Suppression of stem cell proliferation would prevent the epithelial cell turnover required for healing mucosal injuries. They found that administering butyrate reduced epithelial proliferation in the crypts directly adjacent to colonic ulcers and delayed wound repair. In a separate experiment, they found that using antibiotics to knock down butyrate-producing microbes reduced ulcer size after a colitis insult. Reintroduction of butyrate or butyrate-producing organisms resulted in a return to large ulcers.
In a similar study, Zhang et al. introduced a human-derived butyrate-producing bacterium Anaerostipes hadrus into mice with and without colitis. In healthy mice, A. hadrus elevated levels of buyrate in the lumen, and had no adverse effects. In experimental colitis, mice receiving A. hadrus displayed increased disease activity and greater mortality.5
Another study found that supplementation with the soluble fiber inulin, known to stimulate the production of butyrate, resulted in increased pro-inflammatory gene expression in the colon of IBD-prone (IL-10 knockout) mice. This did not occur in control, wild-type mice.6 Together, these studies suggests that the benefits of butyrate, butyrate producers, and even potentially fermentable fibers, depends on the condition of the gut. These data may also partly explain why some inflammatory bowel disease (IBD) patients seem to improve upon reducing FODMAP intake.7
Low abundance of butyrate producers: cause or consequence?
For years, researchers have observed that patients with IBD have reduced levels of butyrate-producing bacteria8–10. It has been thought that this is a potential cause of disease. While this lack of butyrate producers is certainly representative of dysbiosis, this research outlined above makes me wonder if a lack of butyrate producers is actually a consequence of the disease. In other words, is it possible that an inflamed gut that is not in the condition to tolerate luminal butyrate might select against butyrate producers to protect stem cells and allow proliferation and mucosal repair?
After reviewing the data and having these questions come up in my head, I admit I was surprised by the author’s interpretations of their own findings in the discussion:
“Although [butyrate] negatively affects wound repair in the short term, it may actually benefit the host in the long-term. By suppressing the rapid expansion of stem cells following mucosal damage of the epithelium, butyrate may prevent stem cells from dividing while in direct contact with genotoxic luminal contents.”
While this is certainly possible, I think this may be representative of what often happens in science: we get caught up in our own hypotheses and what we think we know to be true. Butyrate has been considered beneficial for quite some time, so it’s hard to wrap our heads around a new paradigm: one in which butyrate could be detrimental in cases of impaired gut integrity. Of course, it’d be nice to have more than just these few studies to rely on. So let’s look at a few others…
Ulcerative colitis patients have impaired butyrate oxidation
Evidence from in vitro and in vivo studies suggests that oxidation of butyrate is impaired in ulcerative colitis (UC). De Preter et al. found that the average butyrate oxidation rate in UC biopsies was reduced to almost half that of the control biopsies. (When stratified based on severity, butyrate oxidation rate in inactive and mild disease was no different than controls, but butyrate oxidation rate in moderate and severe disease was significantly reduced. Interestingly, there was also a huge variability in butyrate oxidation among controls.)11
When you look at studies of butyrate kinetics, it becomes abundantly clear that the concentration of butyrate is not the problem. For both controls and UC biopsies, increasing luminal butyrate concentrations beyond 1mM did not increase butyrate oxidation. It seems that, beyond the 1 mM saturation point, the concentration of butyrate is not the limiting factor. The authors write:
“[…] local application of higher concentrations of butyrate does not overcome the mucosal energy deficiency in UC and […] stimulation of butyrate production through indigestible carbohydrates or butyrate enemas may not be effective in the treatment of this disease.” 11
In a follow-up study, Thibault et al. showed that the decreased butyrate oxidation is a result of reduced butyrate uptake by the inflamed mucosa. Proinflammatory cytokines downregulate expression of MCT1, a transporter of many molecules, including butyrate.12 Kovarik et al. also showed that butyrate has a reduced anti-inflammatory effect in IBD patients.13
Clinical trials, animal studies, and cell culture experiments show mixed results
Indeed, animal studies and human clinical trials have had contradictory results when butyrate enemas or butyrate treatment were tested in ulcerative colitis.
Butyrate enemas in humans:
A beneficial effect of butyrate enemas was found in two small non-placebo controlled studies14,15 in addition to two controlled crossover studies16,17 One small randomized placebo-controlled study in patients with mild to moderate colitis found a beneficial effect of SCFA enemas containing acetate, propionate, and butyrate. 18 However, the largest randomized placebo-controlled trial to date (91 patients) found no therapeutic value of SCFA enemas for colitis, though patients with milder, short episodes of colitis were slightly more likely to benefit19. Another large, randomized placebo-controlled trial (38 patients) found no significant difference between patients receiving saline placebo and butyrate enemas. In fact, 9 of 19 patients saw clinical improvement with the saline enema, while only 7 of 19 patients improved with the butyrate enema over the 6 week study.20 Granted, none of these studies showed an explicit harmful effect of butyrate, but I’d venture to guess that the researchers did not expect one and therefore were not looking for signs of potential mucosal toxicity.
As far as I know, no randomized controlled trials have been done on oral butyrate supplementation in humans.
Animal studies:
What about animal studies? One study found that oral sodium butyrate attenuated colitis in mice.21 However, high concentration butyrate enemas have also been found to induce apoptosis and UC-like lesions in mice22. In a mouse model of colorectal cancer, both inulin and intrarectal butyrate have both been shown to transform the colonic epithelium and increase the formation of tumors.23,24
In mice, chronic orally administered SCFAs induced T cell-mediated ureter tissue inflammation, leading to kidney hydronephrosis, a backup of fluid in the kidney. From the abstract:
“SCFAs, when systemically administered at levels higher than physiological levels, cause dysregulated T cell responses and tissue inflammation in the renal system.” 25
Cell culture:
Notably, several studies have now shown that SCFAs can promote either immune regulation or immune activation, depending on the inflammatory milieu.26,27 In other words, in a state of quiescence, SCFAs promote immune regulation; in a state of inflammation, they promote amplification of the immune response.
Furthermore, butyrate administration at low concentration (2 mM) to a cultured model of an epithelium improves gut barrier integrity by accelerating assembly of tight junction proteins28,29 However, at high concentrations (8 mM), butyrate increases barrier permeability.28
Butyrate also promotes the expression of virulence factors in pathogenic E.coli. These E.coli thus tend to colonize the colon where levels of butyrate are highest.30
Should we ever consider butyrate supplementation?
Possibly. Anecdotally, there are many people who have found benefits of butyrate supplementation, and some patients in the trials I mentioned did see clinical improvement with butyrate enemas. Butyrate certainly has many benefits, as I discussed in part 2 of this series. However, there are currently no double-blind, placebo controlled trials of oral butyrate supplementation in humans that have been performed, so my current thinking is that we need a more nuanced, case-by-case approach.
If you have severe mucosal damage, such as highly active ulcerative colitis, I would tend to advise against butyrate enemas or supplementation. Butyrate might be worth considering for milder cases, for those in remission or with refractory colitis, or for people without GI conditions. Still, it’s important to avoid supra-physiological levels.31
Until recently, the most widely available form of butyrate has been butyrate salts, such as sodium butyrate or calcium/magnesium butyrate. However, butyrate salts may partially dissolve in the mouth. High concentrations of oral butyrate have been associated with periodontitis and may inhibit the proliferation of gingival epithelial cells.32,33
I’ve recently become aware of a new oral supplement formula from Tesseract called ProButyrate that may provide a way to administer lower doses of therapeutic butyrate in a sustained release manner. The molecular trap formulation from Tesseract is specifically targeted for release in the colon and prevents dissolution of butyrate in the oral cavity. It also reduces the dose required to achieve therapeutic effect, with a recommended dose of 600-1200 mg/day, compared to 3-4 g/day for most butyrate salts. I have no affiliation with Tesseract, but look forward to seeing clinical trials on these supplements in the future. If you decide to supplement with butyrate, I believe this is currently the best option available.
Prebiotics: a more physiological butyrate response?
Because of the nature of gut fermentation, prebiotic fibers are also likely to provide a slower and more dispersed release of butyrate in the colon. Our gut anatomy and gut ecology evolved on a diet rich in fermentable fibers, and this is the butyrate precursor that our guts are most accustomed to. Prebiotics also have many benefits beyond butyrate production, including anti-inflammatory effects on the gut epithelium, and the proliferation of beneficial microbes. Still, it’s important to note that even large quantities of prebiotics may be detrimental to healing in cases of severe gut inflammation and mucosal injury.
What If I get a stool test and I have low butyrate? Should I supplement?
Unfortunately, many stool tests that are clinically available use improper sample processing methodology for measuring SCFAs, and therefore cannot be trusted for their accuracy. Furthermore, fecal SCFA levels are not necessarily representative of luminal SCFA concentrations. Stay tuned for part 6 of the SCFA series, where I’ll be addressing this issue in detail.
Takeaways:
- Butyrate can suppress epithelial stem cell proliferation and repair. The normal crypt architecture protects stem cells from high amounts of butyrate exposure.
- Butyrate supplementation should be used with extreme caution in cases of severe gut inflammation and epithelial damage, particularly in active ulcerative colitis. High-dose probiotic and prebiotic supplementation should also be used with caution, as butyrogenic fibers or butyrate-producing bacteria could also potentially exacerbate colitis.
- Small doses of butyrate supplementation may be helpful in those with mild colitis or those without GI issues, but prebiotics likely provide butyrate in more physiologically normal concentrations.
That’s all for now. Next up in the series: acetate and metabolic syndrome!
Sources:
- Stappenbeck, T. S., Wong, M. H., Saam, J. R., Mysorekar, I. U. & Gordon, J. I. Notes from some crypt watchers: regulation of renewal in the mouse intestinal epithelium. Curr. Opin. Cell Biol. 10, 702–709 (1998).
- van der Flier, L. G. & Clevers, H. Stem cells, self-renewal, and differentiation in the intestinal epithelium. Annu. Rev. Physiol. 71, 241–260 (2009).
- Cheng, H. & Leblond, C. P. Origin, differentiation and renewal of the four main epithelial cell types in the mouse small intestine V. Unitarian theory of the origin of the four epithelial cell types. Am. J. Anat. 141, 537–561 (1974).
- Kaiko, G. E. et al. The Colonic Crypt Protects Stem Cells from Microbiota-Derived Metabolites. Cell 165, 1708–1720 (2016).
- Zhang, Q. et al. Accelerated dysbiosis of gut microbiota during aggravation of DSS-induced colitis by a butyrate-producing bacterium. Sci. Rep. 6, (2016).
- Kuo, S.-M., Chan, W.-C. & Hu, Z. Wild-type and IL10-null mice have differential colonic epithelial gene expression responses to dietary supplementation with synbiotic Bifidobacterium animalis subspecies lactis and inulin. J. Nutr. 144, 245–251 (2014).
- Gearry, R. B. et al. Reduction of dietary poorly absorbed short-chain carbohydrates (FODMAPs) improves abdominal symptoms in patients with inflammatory bowel disease-a pilot study. J. Crohns Colitis 3, 8–14 (2009).
- Willing, B. P. et al. A pyrosequencing study in twins shows that gastrointestinal microbial profiles vary with inflammatory bowel disease phenotypes. Gastroenterology 139, 1844–1854.e1 (2010).
- Sokol, H. et al. Low counts of Faecalibacterium prausnitzii in colitis microbiota. Inflamm. Bowel Dis. 15, 1183–1189 (2009).
- Machiels, K. et al. A decrease of the butyrate-producing species Roseburia hominis and Faecalibacterium prausnitzii defines dysbiosis in patients with ulcerative colitis. Gut 63, 1275–1283 (2014).
- De Preter, V. et al. Kinetics of butyrate metabolism in the normal colon and in ulcerative colitis: the effects of substrate concentration and carnitine on the β-oxidation pathway. Aliment. Pharmacol. Ther. 34, 526–532 (2011).
- Thibault, R. et al. Down-Regulation of the Monocarboxylate Transporter 1 Is Involved in Butyrate Deficiency During Intestinal Inflammation. Gastroenterology 133, 1916–1927 (2007).
- Kovarik, J. J. et al. Impaired anti-inflammatory efficacy of n-butyrate in patients with IBD. Eur. J. Clin. Invest. 41, 291–298 (2011).
- Breuer, R. I. et al. Rectal irrigation with short-chain fatty acids for distal ulcerative colitis. Dig. Dis. Sci. 36, 185–187 (1991).
- Steinhart, A. H., Brzezinski, A. & Baker, J. P. Treatment of refractory ulcerative proctosigmoiditis with butyrate enemas. Am. J. Gastroenterol. 89, 179–183 (1994).
- Scheppach, W. et al. Effect of butyrate enemas on the colonic mucosa in distal ulcerative colitis. Gastroenterology 103, 51–56 (1992).
- Lührs, H. et al. Butyrate inhibits NF-kappaB activation in lamina propria macrophages of patients with ulcerative colitis. Scand. J. Gastroenterol. 37, 458–466 (2002).
- Vernia, P. et al. Short-chain fatty acid topical treatment in distal ulcerative colitis. Aliment. Pharmacol. Ther. 9, 309–313 (1995).
- Breuer, R. I. et al. Short chain fatty acid rectal irrigation for left-sided ulcerative colitis: a randomised, placebo controlled trial. Gut 40, 485–491 (1997).
- Steinhart, A. H., Hiruki, T., Brzezinski, A. & Baker, J. P. Treatment of left-sided ulcerative colitis with butyrate enemas: a controlled trial. Aliment. Pharmacol. Ther. 10, 729–736 (1996).
- Vieira, E. L. M. et al. Oral administration of sodium butyrate attenuates inflammation and mucosal lesion in experimental acute ulcerative colitis. J. Nutr. Biochem. 23, 430–436 (2012).
- Ohkusa, T. et al. Induction of experimental ulcerative colitis by Fusobacterium varium isolated from colonic mucosa of patients with ulcerative colitis. Gut 52, 79–83 (2003).
- Pajari, A.-M. et al. Promotion of intestinal tumor formation by inulin is associated with an accumulation of cytosolic beta-catenin in Min mice. Int. J. Cancer 106, 653–660 (2003).
- Belcheva, A. et al. Gut microbial metabolism drives transformation of MSH2-deficient colon epithelial cells. Cell 158, 288–299 (2014).
- Park, J., Goergen, C. J., HogenEsch, H. & Kim, C. H. Chronically Elevated Levels of Short-Chain Fatty Acids Induce T Cell-Mediated Ureteritis and Hydronephrosis. J. Immunol. Baltim. Md 1950 196, 2388–2400 (2016).
- Kim, M. H., Kang, S. G., Park, J. H., Yanagisawa, M. & Kim, C. H. Short-chain fatty acids activate GPR41 and GPR43 on intestinal epithelial cells to promote inflammatory responses in mice. Gastroenterology 145, 396-406.e1–10 (2013).
- Park, J. et al. Short-chain fatty acids induce both effector and regulatory T cells by suppression of histone deacetylases and regulation of the mTOR-S6K pathway. Mucosal Immunol. 8, 80–93 (2015).
- Peng, L., He, Z., Chen, W., Holzman, I. R. & Lin, J. Effects of butyrate on intestinal barrier function in a Caco-2 cell monolayer model of intestinal barrier. Pediatr. Res. 61, 37–41 (2007).
- Peng, L., Li, Z.-R., Green, R. S., Holzman, I. R. & Lin, J. Butyrate Enhances the Intestinal Barrier by Facilitating Tight Junction Assembly via Activation of AMP-Activated Protein Kinase in Caco-2 Cell Monolayers. J. Nutr. 139, 1619–1625 (2009).
- Nakanishi, N. et al. Regulation of virulence by butyrate sensing in enterohaemorrhagic Escherichia coli. Microbiol. Read. Engl. 155, 521–530 (2009).
- Roda, A. et al. A new oral formulation for the release of sodium butyrate in the ileo-cecal region and colon. World J. Gastroenterol. 13, 1079–1084 (2007).
- Tsuda, H., Ochiai, K., Suzuki, N. & Otsuka, K. Butyrate, a bacterial metabolite, induces apoptosis and autophagic cell death in gingival epithelial cells. J. Periodontal Res. 45, 626–634 (2010).
- Ochiai, K. & Kurita-Ochiai, T. Effects of butyric acid on the periodontal tissue. Jpn. Dent. Sci. Rev. 45, 75–82 (2009).
- Burger-van Paassen, N. et al. The regulation of intestinal mucin MUC2 expression by short-chain fatty acids: implications for epithelial protection. Biochem. J. 420, 211–219 (2009).
- Hatayama, H., Iwashita, J., Kuwajima, A. & Abe, T. The short chain fatty acid, butyrate, stimulates MUC2 mucin production in the human colon cancer cell line, LS174T. Biochem. Biophys. Res. Commun. 356, 599–603 (2007).


When they test butyrate enemas, how much of the colon are they emptying in order to retain the enema? If someone has an IBD that ulcerates the ascending colon, how would an enema that only goes into the descending colon help? If they are actually emptying out the whole colon and putting a butyrate enema all the way to the ascending colon, that seems like a highly invasive procedure and would introduce additional risks. The point I am making is that what seems like a simple idea might actually be incredible difficult to test, and differences in study designs would make comparing these tests difficult.
I know that you mentioned low butyrate on fecal samples isn’t always valid. However, with the GI Effects fecal test you recommend—if it shows low butyrate do you believe that finding is likely true?
Thank you for this concise explanation of such a complex topic. I feel I am beginning to grasp the larger picture after mentally digesting any and all Butyrate content I could find of yours on the web!
I am now considering temporary supplementation to advance my gut healing but feel overwhelmed by the very unregulated supplement industry. That being said, while researching, I stumbled across another product on the market (Tributyrin-X) and with it being such a new product I was unable to find much information about it outside of the company endorsements. I am curious if you are familiar with it? It is a liquid form of tributyrin which I have been reading might be a more beneficial delivery system.
I don’t take supplementation lightly and wonder if you had any ideas or additional research you might share on the subject.
Thank you for all that you do and your other product recommendations. Your guidance is truly valued!
In other words, is it possible that an inflamed gut that is not in the condition to tolerate luminal butyrate might select against butyrate producers to protect stem cells and allow proliferation and mucosal repair?
“Although [butyrate] negatively affects wound repair in the short term, it may actually benefit the host in the long-term. By suppressing the rapid expansion of stem cells following mucosal damage of the epithelium, butyrate may prevent stem cells from dividing while in direct contact with genotoxic luminal contents.”
It seems that, beyond the 1 mM saturation point, the concentration of butyrate is not the limiting factor.
In a follow-up study, Thibault et al. showed that the decreased butyrate oxidation is a result of reduced butyrate uptake by the inflamed mucosa. Proinflammatory cytokines downregulate expression of MCT1, a transporter of many molecules, including butyrate.12 Kovarik et al. also showed that butyrate has a reduced anti-inflammatory effect in IBD patients.13
Butyrate enemas in humans:
A beneficial effect of butyrate enemas was found in two small non-placebo controlled studies14,15 in addition to two controlled crossover studies16,17 One small randomized placebo-controlled study in patients with mild to moderate colitis found a beneficial effect of SCFA enemas containing acetate, propionate, and butyrate. 18 However, the largest randomized placebo-controlled trial to date (91 patients) found no therapeutic value of SCFA enemas for colitis, though patients with milder, short episodes of colitis were slightly more likely to benefit19. Another large, randomized placebo-controlled trial (38 patients) found no significant difference between patients receiving saline placebo and butyrate enemas. In fact, 9 of 19 patients saw clinical improvement with the saline enema, while only 7 of 19 patients improved with the butyrate enema over the 6 week study.20 Granted, none of these studies showed an explicit harmful effect of butyrate, but I’d venture to guess that the researchers did not expect one and therefore were not looking for signs of potential mucosal toxicity.
As far as I know, no randomized controlled trials have been done on oral butyrate supplementation in humans.
the takeaway i got is =
“Although [butyrate] negatively affects wound repair in the short term, it may actually benefit the host in the long-term
plus
had a conversation with a leader of a support group of ibs ,and she told me among all avaialble supplements butyrate delivered best results in all people in all ibs conditions
any comments ?
Hello? I have leaky gut probably due cdiff on October, I treated with antibiotic.
A few weeks ago I started a product called permeabiane that contains butyrate (prolonged liberation). I tried once and had a. But of pain in my lower abdomen.
I stopped it and tried again a few days later, then after two days (24 of December 2021) started again the pain so I stopped it. Then I started to have more mushy stools and go to the toilet 4 or 5 times s day with weird colours…
I improved a bit, but now the stools are loose. I went to confirm if cdiff came back and it’s not cdiff
So I’m not sure what happened with me when I was on the butyrate supplement that upset me in that way.
Please, Any idea? I’m very worried and with a lot of anxiety.
Thank you
Hello,
I’m currently taking Bio Complete 3 by Gundry MD. Your article has concerned me and I need to know if I’m risking anything taking this supplement. I do not suffer from ulcers. I’m simply focused on healing my gut and reducing NFLD. I’m sure that I suffer from leaky gut. I do not want to do more harm than good while trying to improve my gut health.
Thanks for this very helpful article! Just found your blog, I’m a new fan :)
Thanks for your comment, Jeremy! Glad you found it helpful – I’ll be doing an update on this one soon to include some clinical studies on butyrate!
There is just one problem. A large percent of people with intestinal conditions feel better with butyrate supplementation. That is my experience as well.
That would not be the case if it was causing harm.
I’ve read comments from people who didn’t find a particular butyrate to help of help much, usually sodium butyrate rather than the better ones like tributyrate. But I don’t think I’ve seen anyone saying it made them worse.
Hello,
I like reading your blog, it helped me a lot.
I have tried to take 2g of Sodium Butyrate, because 1g of it improved my condition.
Unfortunately with 2g I had adverse effects like depression, anxiety and bloated stomach.
Is this what you describe as “excess butyrate” or how much orally taken butyrate is too much? I try to understand the mechanism behind this reaction.
Thank you.
Hi Lucy!
Did you ever post Part 6 or 7 of the series? I’ve been enjoying this so much!!!
Hopefully :-)
Hello! Most forms of butyrate supplementation are absorbed very early in the GI tract. I think I’ve read about butyrate reversing liver pathology (no time to find source right now). I’ve often wondered what happens to butyrate absorbed early in the stomach and small intestine. What does it “do?” Is it good or bad? Science talks a lot about butyrate in the colon, but what about the effects of butyrate that never reaches the colon or distal/mid-distal small intestine? I’ve never found a good schematic for what happens to absorbed butyrate prior to the colon.
As a mom who recently had a daughter throw up on her, my olfactory system thinks it’s keenly aware that there must be butyrate in the stomach. Which bacteria are responsible for that? How much is there being made? What happens to it? Is there a systemic response to butyrate?
Hi Terri! I think this is the study you are referring to regarding butyrate and liver pathology. Very interesting stuff! I may have to update the article to include this one at some point.
https://www.ncbi.nlm.nih.gov/pubmed/28223285
I agree that there is not much literature on the stomach and small intestine. However, in a typical gut in a person that is not supplementing, the majority of butyrate is produced by bacterial fermentation in the colon. One study in humans found that “total SCFA concentration (mmol/kg) was low in the terminal ileum at 13 +/- 6 but high in all regions of the colon ranging from 131 +/- 9 in the caecum to 80 +/- 11 in the descending colon.”
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1433442/
So, I don’t think there was butyrate in your daughter’s stomach, unless she was supplementing with butyrate!
Also, butyrate is transported into intestinal epithelial cells primarily through monocarboxylate transporter 1 (MCT1), which is expressed about 20-fold higher in the colon compared to the small intestine. I am not aware of any other mechanism that has been identified by which butyrate is absorbed in the stomach or small intestine in significant quantities – although it’s possible we may discover one in the future!
So, with supplements such as sodium butyrate, where butyrate is liberated before reaching the small intestine, what happens? The butyrate receptors FFAR2, FFAR3, and GPR109a are expressed in the small intestine, so butyrate could certainly have some important signaling functions there. But if there aren’t any transporters or other means of absorbing butyrate until reaching the colon, you’d likely be stuck with high luminal butyrate concentrations in the small intestine, which might cause damage to crypts there as well. This is why I tend to think that colon-targeted butyrate supplements or prebiotic fibers are the best way to go, since butyrate will be produced in smaller quantities where it is needed and actively metabolized.
Best,
Lucy